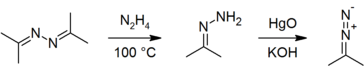Azine
Azines are a functional class of organic compounds, formed from the condensation reaction of two equivalents of an aldehyde or ketone with one equivalent of hydrazine.[1][2] They may be further classified as aldazines or ketazines, depending on the nature of the carbonyl compound.[3][4]
Preparation
Azines may be prepared by the direct reaction of a carbonyl compound with hydrazine hydrate:[5] the reaction is exothermic.[6] The usual method of industrial production is the Pechiney-Ugine-Kuhlmann process, starting from the ketone, ammonia and hydrogen peroxide.[7][8]
Reactions and uses
Azines have been used as precursors to hydrazones[5][9] and diazo compounds.[9][10][11]
Azines are also important intermediates in the industrial production of hydrazine hydrate by the Bayer hydrazine process[12][13] or the Pechiney-Ugine-Kuhlmann process.[7][8] They have been also used as sources of hydrazine produced in situ, for example in the production of herbicide precursor 1,2,4-triazole.[14]
The coordination chemistry of azines (as ligands) has also been studied.[15][16][17] Acetone is used to derivatize hydrazine, through formation of acetone azine, for analysis by gas chromatography: the method has been used to determine trace levels of hydrazine in drinking water[18] and pharmaceuticals.[19]
Nomenclature
Azines may be named by substitutive or functional class nomenclature.[20][21] In functional class nomenclature, the functional modifier "azine" is appended to the name of the carbonyl compound: hence, "acetone azine".[20] In older nomenclature, the functional class name "ketazine" has been used with the names of the hydrocarbyl substituents: e.g., "methyl ethyl ketazine". In substitutive nomenclature, azines are named as derivatives of hydrazine: hence, "diisopropylidenehydrazine".[20] In the presence of groups of higher seniority, the prefixes "hydrazinylidene" and "hydrazinediylidene" are used.[22]
Unsymmetrical azines, that is compounds of the type X=N–N=Y with X ≠ Y, are not named as azines: in the absence of other functional groups having higher seniority, they can be named as substituted hydrazones.[23]
References
- ↑ Glossary of class names of organic compounds and reactivity intermediates based on structure (IUPAC Recommendations 1995). Pure Appl. Chem. 1995, 67 (8-9), 1307–75 at 1321. DOI: 10.1351/pac199567081307.
- ↑ azines, <http://goldbook.iupac.org/A00557.html> (accessed 2 July 2010), Compendium of Chemical Terminology Internet edition; International Union of Pure and Applied Chemistry (IUPAC).
- ↑ Glossary of class names of organic compounds and reactivity intermediates based on structure (IUPAC Recommendations 1995). Pure Appl. Chem. 1995, 67 (8-9), 1307–75 at 1312, 1348. DOI: 10.1351/pac199567081307.
- ↑ aldazines, <http://goldbook.iupac.org/A00207.html> (accessed 2 July 2010), Compendium of Chemical Terminology Internet edition; International Union of Pure and Applied Chemistry (IUPAC). ketazines, <http://goldbook.iupac.org/K03377.html> (accessed 2 July 2010), Compendium of Chemical Terminology Internet edition; International Union of Pure and Applied Chemistry (IUPAC).
- ↑ 5.0 5.1 Day, A. C.; Whiting, M. C. Acetone hydrazone. Org. Synth. 1970, 50, 3, <http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv6p0010>; Coll. Vol., 6, 10.
- ↑ Gilbert, E. C. Studies on Hydrazine. The Hydrolysis of Dimethylketazine and the Equilibrium between Hydrazine and Acetone. J. Am. Chem. Soc. 1929, 51 (11), 3394–3409. DOI: 10.1021/ja01386a032.
- ↑ 7.0 7.1 Schirmann, Jean-Pierre; Combroux, Jean; Delavarenne, Serge Yvon (Produits Chimiques Ugine Kuhlmann) Method for preparing azines and hydrazones. US Patent 3972878, issued 3 August 1976. Schirmann, Jean-Pierre; Tellier, Pierre; Mathais, Henri, et al. (Produits Chimiques Ugine Kuhlmann) Process for the preparation of hydrazine compounds. US Patent 3978049, issued 31 August 1976. Schirmann, Jean-Pierre; Combroux, Jean; Delavarenne, Serge Yvon (Produits Chimiques Ugine Kuhlmann) Method for making azines. US Patent 4093656, issued 6 June 1978.
- ↑ 8.0 8.1 Schirmann, Jean-Pierre; Combroux, Jean; Delavarenne, Serge Y. (Atochem) Preparation of a concentrated aqueous solution of hydrazine hydrate. US Patent 4724133, issued 9 February 1988.
- ↑ 9.0 9.1 Staudinger, H.; Gaule, Alice Vergleich der Stickstoff-Abspaltung bei verschiedenen aliphatischen Diazoverbindungen. Ber. Dtsch. Chem. Ges. 1916, 49 (2), 1897–1918. DOI: 10.1002/cber.19160490245.
- ↑ Day, A. C.; Raymond, P.; Southam, R. M.; Whiting, M. C. The preparation of secondary aliphatic diazo-compounds from hydrazones. J. Chem. Soc. C 1966, 467–69. DOI: 10.1039/J39660000467.
- ↑ Andrews, S. D.; Day, A. C.; Raymond, P.; Whiting, M. C. 2-Diazopropane. Org. Synth. 1970, 50, 27, <http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=cv6p0392>; Coll. Vol., 6, 392.
- ↑ Eichenhofer, Kurt-Wilhelm; Schliebs, Reinhard (Bayer) Production of ketazines. US Patent 3965097, issued 22 June 1976.
- ↑ Holleman, A. F.; Wiberg, E. Inorganic Chemistry; Academic Press: San Diego, 2001; p 619. ISBN 0-12-352651-5.
- ↑ Nagata, Nobuhiro; Nishizawa, Chiharu; Kurai, Toshikiyo (Mitsubishi Gas Chemical Co.) Method of producing 1,2,4-triazole. US Patent 6002015, issued 14 December 1999.
- ↑ Gudkova, A. S.; Reutov, O. A.; Aleinikova, M. Ya. Izv. Akad. Nauk SSSR, Otdel. Khim. Nauk 1962 (8), 1382–87; Reactions of hydrazones and azines with metal salts 4. Reactions of azines of aldehydes and ketones with cupric salts. Russ. Chem. Bull. (Transl.) 1962, 11 (8), 1298–1302. DOI: 10.1007/BF00907973.
- ↑ Gudkova, A. S.; Aleinikova, M. Ya.; Reutov, O. A. Izv. Akad. Nauk SSSR, Ser. Khim. 1966 (5), 844–48; Reactions of hydrazones and azines with metal salts 5. Reactions of hydrazones and azines with mercuric halides. Russ. Chem. Bull. (Transl.) 1966, 15 (5), 807–11. DOI: 10.1007/BF00849376.
- ↑ King, Fiona; Nicholls, David Complex of titanium halides with acetone azine and its isomer 3,5,5-trimethyl-pyrazoline. Inorg. Chim. Acta 1978, 28, 55–58. DOI: 10.1016/S0020-1693(00)87413-7.
- ↑ Davis, William E., II; Li, Yongtao Analysis of hydrazine in drinking water by isotope dilution gas chromatography/tandem mass spectrometry with derivatization and liquid-liquid extraction. Anal. Chem. 2008, 80 (14), 5449–53. DOI: 10.1021/ac702536d.
- ↑ Sun, Mingjiang; Bai, Lin; Liu, David Q. A generic approach for the determination of trace hydrazine in drug substances using in situ derivatization-headspace GC–MS. J. Pharm. Biomed. Anal. 2009, 49 (2), 529–33. DOI: 10.1016/j.jpba.2008.11.009.
- ↑ 20.0 20.1 20.2 Rule R-5.6.6.3. In A Guide to IUPAC Nomenclature of Organic Compounds; IUPAC Recommendations 1993; Blackwell Science: Oxford, 1993; p 105. ISBN 0-632-03488-2, <http://www.acdlabs.com/iupac/nomenclature/93/r93_469.htm>.
- ↑ Draft Rule P-68.3.1.2.3. In Nomenclature of Organic Chemistry; IUPAC Provisional Recommendations 2004; IUPAC, 2004, <http://old.iupac.org/reports/provisional/abstract04/favre_310305.html>.
- ↑ Draft Rule P-53.6.2. In Nomenclature of Organic Chemistry; IUPAC Provisional Recommendations 2004; IUPAC, 2004, <http://old.iupac.org/reports/provisional/abstract04/favre_310305.html>.
- ↑ Rule C-923.2. In Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F, and H; IUPAC Recommendations 1979; Pergamon: Oxford, 1979. ISBN 0-08022-369-9, <http://www.acdlabs.com/iupac/nomenclature/79/r79_623.htm>.
| Error creating thumbnail: Unable to save thumbnail to destination |
This page is currently licensed under the Creative Commons Attribution 3.0 Unported license and any later versions of that license. |