Difference between revisions of "Air-free technique"
Rifleman 82 (talk | contribs) (New page: '''Air-free techniques''' refer to a range of manipulations in the chemistry laboratory for the handling of compounds that are air-sensitive. These techniques prevent the compounds...) |
Physchim62 (talk | contribs) (Reimported from http://en.wikipedia.org/w/index.php?title=Air-free_technique&oldid=281851600 for copyright reasons) |
||
| Line 1: | Line 1: | ||
| − | '''Air-free techniques''' refer to a range of manipulations in the chemistry [[laboratory]] for the handling of compounds that are [[air-sensitive]]. These techniques prevent the compounds from reacting with components of [[air]], usually [[water]] and [[oxygen]]; less commonly [[carbon dioxide]] and [[nitrogen]]. A common theme among these techniques is the use of a high [[vacuum]] to remove air, and the use of an [[inert gas]]: preferably [[argon]], but often [[nitrogen]]. | + | '''Air-free techniques''' refer to a range of manipulations in the chemistry [[laboratory]] for the handling of [[Compound (chemistry)|compounds]] that are [[air-sensitive]]. These techniques prevent the compounds from reacting with components of [[air]], usually [[water]] and [[oxygen]]; less commonly [[carbon dioxide]] and [[nitrogen]]. A common theme among these techniques is the use of a high [[vacuum]] to remove air, and the use of an [[inert gas]]: preferably [[argon]], but often [[nitrogen]]. |
| − | The two most common types of air-free technique involve the use of a [[glovebox]] and a [[Schlenk line]]. In both methods, glassware are pre-dried in ovens prior to use. They may be flame-dried to remove adsorbed water. Prior to coming into an inert atmosphere, vessels are further dried by ''purge-and-refill'' — the vessel is subjected to a vacuum to remove gases and water, and then refilled with inert gas. This cycle is usually repeated | + | The two most common types of air-free technique involve the use of a [[glovebox]] and a [[Schlenk line]]. In both methods, glassware (often [[Schlenk tube]]s) are pre-dried in ovens prior to use. They may be flame-dried to remove adsorbed water. Prior to coming into an inert atmosphere, vessels are further dried by ''purge-and-refill'' — the vessel is subjected to a vacuum to remove gases and water, and then refilled with inert gas. This cycle is usually repeated three times or the vacuum is applied for an extend period of time. One of the differences between the use of a glovebox and a Schlenk line is where the ''purge-and-refill'' cycle is applied. When using a glovebox the ''purge-and-refill'' is applied to an [[airlock]] attached to the glovebox, commonly called the "port" or "ante-chamber". In contrast when using a Schlenk line the ''purge-and-refill'' is applied directly to the reaction vessel through a hose or ground glass joint that is connected to the manifold.<ref>Duward F. Shriver and M. A. Drezdzon "The Manipulation of Air-Sensitive Compounds" 1986, J. Wiley and Sons: New York. ISBN 0-471-86773-X.</ref> |
==Glovebox== | ==Glovebox== | ||
[[Image:Glovebox.jpg|thumb|An ordinary glovebox, showing the two gloves for manipulation, with airlock on the right.]] | [[Image:Glovebox.jpg|thumb|An ordinary glovebox, showing the two gloves for manipulation, with airlock on the right.]] | ||
| − | The most straightforward type of air-free technique is the use of a [[glovebox]]. A [[glove bag]] uses the same idea, but is usually a poorer substitute because it is more difficult to purge, and less well sealed. | + | The most straightforward type of air-free technique is the use of a [[glovebox]]. A [[glove bag]] uses the same idea, but is usually a poorer substitute because it is more difficult to purge, and less well sealed. Inventive ways of accessing items beyond the reach of the gloves exist, such as the use of tongs and strings. The main drawbacks to using a glovebox are the cost of the glovebox itself, and limited dexterity wearing the gloves. |
| − | + | In the glovebox, conventional laboratory equipment can often be set up and manipulated, despite the need to handle the apparatus with the gloves. By providing a sealed but recirculating atmosphere of the inert gas, the glove box necessitates few other precautions. Cross contamination of samples due to poor technique is also problematic, especially where a glovebox is shared between workers using differing reagents; [[Volatility (chemistry)|volatile]] ones in particular. | |
| − | + | Two styles have evolved in the use of gloveboxes for [[synthetic chemistry]]. In a more conservative mode, they are used solely to store, weigh, and transfer air-sensitive [[reagents]]. Reactions are thereafter carried out using Schlenk techniques. The gloveboxes are thus only used for the most air-sensitive stages in an experiment. In their more liberal use, gloveboxes are used for the entire synthetic operations including reactions in solvents, work-up, and preparation of samples for spectroscopy. | |
| + | |||
| + | Not all reagents and solvents are acceptable for use in the glovebox, although again, different laboratories adopt different cultures. The "box atmosphere" is usually continuously deoxygenated over a copper catalyst. Certain volatile chemicals such as halogenated compounds and especially strongly coordinating species such as [[phosphines]] and [[thiol]]s can be problematic because they irreversibly poison the copper catalyst. Because of this problem, many experimentalists choose to handle such compounds using Schlenk techniques. In the more liberal use of gloveboxes, it is accepted that the copper catalyst will require more frequent replacement but this cost is considered to be an acceptable trade-off for the efficiency of conducting an entire synthesis within a protected environment | ||
==Schlenk line== | ==Schlenk line== | ||
| + | {{main|Schlenk line}} | ||
[[Image:Double vac line front view.jpg|thumb|A Schlenk line with four ports.]] | [[Image:Double vac line front view.jpg|thumb|A Schlenk line with four ports.]] | ||
| − | The other main | + | The other main technique for the preparation and handing of air-sensitive compounds are associated with the use of a Schlenk line. The main techniques include: |
* counterflow additions, where air-stable [[reagent]]s are added to the reaction vessel against a flow of inert gas. | * counterflow additions, where air-stable [[reagent]]s are added to the reaction vessel against a flow of inert gas. | ||
| − | * the use of rubber [[ | + | * the use of [[syringes]] and rubber [[septa]] to transfer liquids and solutions |
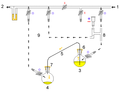
| − | * | + | * [[cannula transfer]], where liquids or solutions of air-sensitive reagents are transferred between different vessels stoppered with septa using a long thin tube known as a cannula. Liquid flow is achieved via vacuum or inert gas pressure.<ref>Brown, H. C. “Organic Syntheses via Boranes” John Wiley & Sons, Inc. New York: 1975. ISBN 0-471-11280-1.</ref>[[Image:Aldolrxnpic.jpg|thumb|right|A cannula is used to transfer [[Tetrahydrofuran|THF]] from the flask on the right to the flask on the left.]] |
| − | |||
| − | |||
| − | |||
| − | |||
| − | + | Glassware are usually connected via tightly-fitting and greased [[ground glass joint]]s. Round bends of [[glass tubing]] with ground glass joints may be used to adjust the orientation of various vessels. Filtrations may be accomplished by dedicated equipment. | |
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
==Associated preparations== | ==Associated preparations== | ||
| − | Commercially available purified inert gas (argon or nitrogen) is adequate for most purposes. However, for certain applications, it is necessary to further remove water and oxygen. This can be accomplished by piping the inert gas line through a heated column of copper [[catalyst]], which converts the oxygen to | + | Commercially available purified inert gas (argon or nitrogen) is adequate for most purposes. However, for certain applications, it is necessary to further remove water and oxygen. This additional purification can be accomplished by piping the inert gas line through a heated column of copper [[catalyst]], which converts the oxygen to copper oxide. Water is removed by piping the gas through a column of desiccant such as [[phosphorus pentoxide]] or molecular sieves. |
| − | Air- and water-free solvents are also necessary. If high-purity solvents are available in nitrogen-purged [[Winchester (bottle)|Winchesters]], they can be brought directly into the glovebox. For use with Schlenk technique, they can be quickly poured into [[Schlenk flask]]s charged with molecular sieves, and [[degasification|degassed]]. | + | Air- and water-free solvents are also necessary. If high-purity solvents are available in nitrogen-purged [[Winchester (bottle)|Winchesters]], they can be brought directly into the glovebox. For use with Schlenk technique, they can be quickly poured into [[Schlenk flask]]s charged with molecular sieves, and [[degasification|degassed]]. More typically, solvent is dispensed directly from a still or solvent purification column. |
===Degassing=== | ===Degassing=== | ||
{{see also|degasification}} | {{see also|degasification}} | ||
| − | Two procedures for degassing are common. The first is known as ''freeze-pump-thaw'' — the solvent is frozen under [[liquid nitrogen]], and a vacuum is applied. Thereafter, the stopcock is closed and the solvent is thawed in warm water, allowing trapped bubbles of gas escape.<ref>{{cite web | + | Two procedures for degassing are common. The first is known as ''freeze-pump-thaw'' — the solvent is frozen under [[liquid nitrogen]], and a vacuum is applied. Thereafter, the stopcock is closed and the solvent is thawed in warm water, allowing trapped bubbles of gas to escape.<ref>{{cite web | publisher = [[University of Houston]] | url = http://www.nanostructure.uh.edu/Forms/ | title = Procedure for Degassing of Liquids using Freeze-Pump-Thaw}}</ref> |
| − | The second procedure is to simply subject the solvent to a vacuum. Stirring or mechanical agitation using | + | The second procedure is to simply subject the solvent to a vacuum. Stirring or mechanical agitation using an [[ultrasonicator]] is useful. Dissolved gases evolve first; once the solvent starts to evaporate, noted by condensation outside the flask walls, the flask is refilled with inert gas. Both procedures are repeated three times. |
===Drying=== | ===Drying=== | ||
[[Image:Distillation of dry and oxygen-free toluene.jpg|thumb|After being refluxed with sodium and benzophenone to remove oxygen and water, toluene is distilled under inert gas into a receiving flask.]] | [[Image:Distillation of dry and oxygen-free toluene.jpg|thumb|After being refluxed with sodium and benzophenone to remove oxygen and water, toluene is distilled under inert gas into a receiving flask.]] | ||
| − | Solvent are traditionally purified by [[distillation]] over an appropriate [[desiccant]] under an inert atmosphere. The use of [[ | + | Solvent are traditionally purified by [[distillation]] over an appropriate [[desiccant]] under an inert atmosphere. The main problem with the use of sodium as a desiccant (below its melting point) is associated with the slow rate of reaction between a solid and a solution. When however, the desiccant is soluble, the speed of drying is much higher. [[Benzophenone]] is often used to generate such a soluble drying agent. An advantage to this application is the intense blue color of the [[ketyl]] [[radical anion]]. Thus, sodium/benzophenone can be used as an indicator of air-free and moisture-free conditions in the purification of solvents by distillation. <ref>{{cite web | author = Nathan L. Bauld | publisher = [[University of Texas]] | year = 2001 | title = Unit 6: Anion Radicals | url = http://research.cm.utexas.edu/nbauld/unit6_anrad.htm}}</ref><ref>{{cite book | author = W. L. F. Armarego and C. Chai | title = Purification of laboratory chemicals | year = 2003 | publisher = Butterworth-Heinemann | city = Oxford | isbn = 0750675713}}</ref> |
| − | However, distillation stills are fire hazards and are increasingly being replaced by alternative solvent-drying systems. Particularly popular is the filtration of deoxygenated solvents through columns filled | + | However, distillation stills are fire hazards and are increasingly being replaced by alternative solvent-drying systems. Particularly popular is the filtration of deoxygenated solvents through columns filled with activated [[alumina]]. <ref>{{cite journal | author = Pangborn, A. B.; Giardello, M. A.; Grubbs, R. H.; Rosen, R. K. and Timmers, F. J. | title = Safe and Convenient Procedure for Solvent Purification | journal = [[Organometallics]] | year = 1996 | volume = 15 | issue = 5 | pages = 1518–20 | doi = 10.1021/om9503712}}</ref> |
| + | |||
| + | Drying of solids can be brought about by storing the solid over a drying agent such as [[phosphorus pentoxide]] ({{chem|P|2|O|5}}) or [[silica gel]], storing in a drying oven/vacuum-drying oven, heating under a high vacuum or in a [[drying pistol]], or to remove trace amounts of water, simply storing the solid in a glove box that has a dry atmosphere. | ||
==Alternatives== | ==Alternatives== | ||
| Line 54: | Line 49: | ||
==References== | ==References== | ||
<references/> | <references/> | ||
| + | |||
| + | ==See also== | ||
| + | *[[Sparging (chemistry)]] | ||
| + | *[[Degasification]] | ||
==External links== | ==External links== | ||
| − | * {{cite web | author = Rob Toreki | title = Glove Boxes | url = http://www.ilpi.com/inorganic/glassware/glovebox.html | work = The Glassware Gallery | publisher = Interactive Learning Paradigms Incorporated | date = 24 | + | * {{cite web | author = Rob Toreki | title = Glove Boxes | url = http://www.ilpi.com/inorganic/glassware/glovebox.html | work = The Glassware Gallery | publisher = Interactive Learning Paradigms Incorporated | date = 2004-05-24}} |
| − | * {{cite web | author = Rob Toreki | title = Schlenk Lines and Vacuum Lines | url = http://www.ilpi.com/inorganic/glassware/vacline.html | work = The Glassware Gallery | publisher = Interactive Learning Paradigms Incorporated | date = 25 | + | * {{cite web | author = Rob Toreki | title = Schlenk Lines and Vacuum Lines | url = http://www.ilpi.com/inorganic/glassware/vacline.html | work = The Glassware Gallery | publisher = Interactive Learning Paradigms Incorporated | date = 2004-05-25}} |
* {{cite web | author = Jürgen Heck | title = The Integrated Synthesis Course: Schlenk Technique | url = http://www.kok.chembio.ntnu.no/chemtalk/Artikler/Schlenk%20technique/ISP-Labcourse.pdf | publisher = [[University of Hamburg]] | format = reprint at Norwegian University of Science and Technology}} | * {{cite web | author = Jürgen Heck | title = The Integrated Synthesis Course: Schlenk Technique | url = http://www.kok.chembio.ntnu.no/chemtalk/Artikler/Schlenk%20technique/ISP-Labcourse.pdf | publisher = [[University of Hamburg]] | format = reprint at Norwegian University of Science and Technology}} | ||
| − | * {{cite web | title = Handling and Storage of Air-Sensitive Reagents | publisher = [[Sigma-Aldrich]] | url = http://www.sigmaaldrich.com/aldrich/bulletin/al_techbull_al134.pdf}} | + | * {{cite web | title = AL-134: Handling and Storage of Air-Sensitive Reagents | work = Technical Bulletin | publisher = [[Sigma-Aldrich]] | url = http://www.sigmaaldrich.com/aldrich/bulletin/al_techbull_al134.pdf}} |
| + | |||
| + | == Gallery == | ||
| + | <gallery> | ||
| + | Image:Perkin_triangle_distillation_apparatus.svg | [[Perkin triangle]]: Air-sensitive distillations | ||
| + | Image:Air-free_filtration.svg | Air-free filtration | ||
| + | Image:Air-free sublimation.png | Air-free sublimation | ||
| + | Image:Air sensitive cannula -intra-bleed valve.png | Cannula: intra-bleed valve | ||
| + | Image:Air sensitive cannula -extra-bleed valve.png | Cannula: extra-bleed valve | ||
| + | Image:Air sensitive cannula - no bleed valve.png | Cannula: (Simple) no bleed valve | ||
| + | Image:air sensitive cannula - two manifold system.png | Cannula: two manifold system | ||
| + | Image:Air sensitive cannula - syringe valve.png | Cannula: syringe valve | ||
| + | Image:NMRtubeTeflontap.png | Teflon tap for air-sensitive NMR samples | ||
| + | </gallery> | ||
| + | |||
| + | [[Category:Air-free techniques|*]] | ||
| − | + | {{Imported from Wikipedia|name=Air-free technique|id=281851600}} | |
Latest revision as of 09:17, 1 August 2009
Air-free techniques refer to a range of manipulations in the chemistry laboratory for the handling of compounds that are air-sensitive. These techniques prevent the compounds from reacting with components of air, usually water and oxygen; less commonly carbon dioxide and nitrogen. A common theme among these techniques is the use of a high vacuum to remove air, and the use of an inert gas: preferably argon, but often nitrogen.
The two most common types of air-free technique involve the use of a glovebox and a Schlenk line. In both methods, glassware (often Schlenk tubes) are pre-dried in ovens prior to use. They may be flame-dried to remove adsorbed water. Prior to coming into an inert atmosphere, vessels are further dried by purge-and-refill — the vessel is subjected to a vacuum to remove gases and water, and then refilled with inert gas. This cycle is usually repeated three times or the vacuum is applied for an extend period of time. One of the differences between the use of a glovebox and a Schlenk line is where the purge-and-refill cycle is applied. When using a glovebox the purge-and-refill is applied to an airlock attached to the glovebox, commonly called the "port" or "ante-chamber". In contrast when using a Schlenk line the purge-and-refill is applied directly to the reaction vessel through a hose or ground glass joint that is connected to the manifold.[1]
Contents
Glovebox
The most straightforward type of air-free technique is the use of a glovebox. A glove bag uses the same idea, but is usually a poorer substitute because it is more difficult to purge, and less well sealed. Inventive ways of accessing items beyond the reach of the gloves exist, such as the use of tongs and strings. The main drawbacks to using a glovebox are the cost of the glovebox itself, and limited dexterity wearing the gloves.
In the glovebox, conventional laboratory equipment can often be set up and manipulated, despite the need to handle the apparatus with the gloves. By providing a sealed but recirculating atmosphere of the inert gas, the glove box necessitates few other precautions. Cross contamination of samples due to poor technique is also problematic, especially where a glovebox is shared between workers using differing reagents; volatile ones in particular.
Two styles have evolved in the use of gloveboxes for synthetic chemistry. In a more conservative mode, they are used solely to store, weigh, and transfer air-sensitive reagents. Reactions are thereafter carried out using Schlenk techniques. The gloveboxes are thus only used for the most air-sensitive stages in an experiment. In their more liberal use, gloveboxes are used for the entire synthetic operations including reactions in solvents, work-up, and preparation of samples for spectroscopy.
Not all reagents and solvents are acceptable for use in the glovebox, although again, different laboratories adopt different cultures. The "box atmosphere" is usually continuously deoxygenated over a copper catalyst. Certain volatile chemicals such as halogenated compounds and especially strongly coordinating species such as phosphines and thiols can be problematic because they irreversibly poison the copper catalyst. Because of this problem, many experimentalists choose to handle such compounds using Schlenk techniques. In the more liberal use of gloveboxes, it is accepted that the copper catalyst will require more frequent replacement but this cost is considered to be an acceptable trade-off for the efficiency of conducting an entire synthesis within a protected environment
Schlenk line
The other main technique for the preparation and handing of air-sensitive compounds are associated with the use of a Schlenk line. The main techniques include:
- counterflow additions, where air-stable reagents are added to the reaction vessel against a flow of inert gas.
- the use of syringes and rubber septa to transfer liquids and solutions
- cannula transfer, where liquids or solutions of air-sensitive reagents are transferred between different vessels stoppered with septa using a long thin tube known as a cannula. Liquid flow is achieved via vacuum or inert gas pressure.[2]
 A cannula is used to transfer THF from the flask on the right to the flask on the left.
A cannula is used to transfer THF from the flask on the right to the flask on the left.
Glassware are usually connected via tightly-fitting and greased ground glass joints. Round bends of glass tubing with ground glass joints may be used to adjust the orientation of various vessels. Filtrations may be accomplished by dedicated equipment.
Associated preparations
Commercially available purified inert gas (argon or nitrogen) is adequate for most purposes. However, for certain applications, it is necessary to further remove water and oxygen. This additional purification can be accomplished by piping the inert gas line through a heated column of copper catalyst, which converts the oxygen to copper oxide. Water is removed by piping the gas through a column of desiccant such as phosphorus pentoxide or molecular sieves.
Air- and water-free solvents are also necessary. If high-purity solvents are available in nitrogen-purged Winchesters, they can be brought directly into the glovebox. For use with Schlenk technique, they can be quickly poured into Schlenk flasks charged with molecular sieves, and degassed. More typically, solvent is dispensed directly from a still or solvent purification column.
Degassing
- See also: degasification
Two procedures for degassing are common. The first is known as freeze-pump-thaw — the solvent is frozen under liquid nitrogen, and a vacuum is applied. Thereafter, the stopcock is closed and the solvent is thawed in warm water, allowing trapped bubbles of gas to escape.[3]
The second procedure is to simply subject the solvent to a vacuum. Stirring or mechanical agitation using an ultrasonicator is useful. Dissolved gases evolve first; once the solvent starts to evaporate, noted by condensation outside the flask walls, the flask is refilled with inert gas. Both procedures are repeated three times.
Drying
Solvent are traditionally purified by distillation over an appropriate desiccant under an inert atmosphere. The main problem with the use of sodium as a desiccant (below its melting point) is associated with the slow rate of reaction between a solid and a solution. When however, the desiccant is soluble, the speed of drying is much higher. Benzophenone is often used to generate such a soluble drying agent. An advantage to this application is the intense blue color of the ketyl radical anion. Thus, sodium/benzophenone can be used as an indicator of air-free and moisture-free conditions in the purification of solvents by distillation. [4][5]
However, distillation stills are fire hazards and are increasingly being replaced by alternative solvent-drying systems. Particularly popular is the filtration of deoxygenated solvents through columns filled with activated alumina. [6]
Drying of solids can be brought about by storing the solid over a drying agent such as phosphorus pentoxide (P2O5) or silica gel, storing in a drying oven/vacuum-drying oven, heating under a high vacuum or in a drying pistol, or to remove trace amounts of water, simply storing the solid in a glove box that has a dry atmosphere.
Alternatives
Both these techniques require rather expensive equipment. Where air-free requirements are not as stringent, other techniques exist. For example, for preparing Grignard reagents which hydrolyze on exposure to water, it is usually sufficient to connect a guard tube filled with calcium chloride to the reflux condenser to prevent moisture contamination.
In situ desiccants such as molecular sieves, or the removal of water by azeotropic distillation can also be used.
References
- ↑ Duward F. Shriver and M. A. Drezdzon "The Manipulation of Air-Sensitive Compounds" 1986, J. Wiley and Sons: New York. ISBN 0-471-86773-X.
- ↑ Brown, H. C. “Organic Syntheses via Boranes” John Wiley & Sons, Inc. New York: 1975. ISBN 0-471-11280-1.
- ↑ Procedure for Degassing of Liquids using Freeze-Pump-Thaw. University of Houston.
- ↑ Nathan L. Bauld (2001). Unit 6: Anion Radicals. University of Texas.
- ↑ W. L. F. Armarego and C. Chai (2003). Purification of laboratory chemicals. Butterworth-Heinemann. ISBN 0750675713.
- ↑ Pangborn, A. B.; Giardello, M. A.; Grubbs, R. H.; Rosen, R. K. and Timmers, F. J. (1996). "Safe and Convenient Procedure for Solvent Purification". Organometallics 15 (5): 1518–20. doi:10.1021/om9503712.
See also
External links
- Rob Toreki (2004-05-24). Glove Boxes. The Glassware Gallery. Interactive Learning Paradigms Incorporated.
- Rob Toreki (2004-05-25). Schlenk Lines and Vacuum Lines. The Glassware Gallery. Interactive Learning Paradigms Incorporated.
- Jürgen Heck. The Integrated Synthesis Course: Schlenk Technique (reprint at Norwegian University of Science and Technology). University of Hamburg.
- AL-134: Handling and Storage of Air-Sensitive Reagents. Technical Bulletin. Sigma-Aldrich.
Gallery
Perkin triangle: Air-sensitive distillations
| Error creating thumbnail: Unable to save thumbnail to destination | |
This page was originally imported from Wikipedia, specifically this version of the article "Air-free technique". Please see the history page on Wikipedia for the original authors. This WikiChem article may have been modified since it was imported. It is licensed under the Creative Commons Attribution–Share Alike 3.0 Unported license. |