Difference between revisions of "Nomenclature of steroids"
Physchim62 (talk | contribs) (→Parent hydride names) |
Physchim62 (talk | contribs) (→Parent hydride names) |
||
| Line 4: | Line 4: | ||
A [[steroid]] is a compound (either naturally occurring or artificial) based on the cyclopenta[''a'']phenanthrene carbon skeleton, partially or completely hydrogenated.<ref name="GoldBook">{{GoldBookRef|title=steroids|file=S06005|accessdate=2010-03-08}}.</ref><ref name="ClassNames">{{IUPAC class names 1995|page=1367}}.</ref> Steroids usually have methyl groups at C-10 and C-13, and often an alkyl group at C-17 (termed a "side chain"). By extension, one or more bond scissions, ring expansions and/or ring contractions of the skeleton may have occurred.<ref name="GoldBook"/><ref name="ClassNames"/> [[Sterol]]s are steroids with a hydroxyl group at C-3.<ref name="ClassNames"/> | A [[steroid]] is a compound (either naturally occurring or artificial) based on the cyclopenta[''a'']phenanthrene carbon skeleton, partially or completely hydrogenated.<ref name="GoldBook">{{GoldBookRef|title=steroids|file=S06005|accessdate=2010-03-08}}.</ref><ref name="ClassNames">{{IUPAC class names 1995|page=1367}}.</ref> Steroids usually have methyl groups at C-10 and C-13, and often an alkyl group at C-17 (termed a "side chain"). By extension, one or more bond scissions, ring expansions and/or ring contractions of the skeleton may have occurred.<ref name="GoldBook"/><ref name="ClassNames"/> [[Sterol]]s are steroids with a hydroxyl group at C-3.<ref name="ClassNames"/> | ||
| − | == | + | ==Fundamental parent structures== |
{| class="wikitable" | {| class="wikitable" | ||
|- | |- | ||
| − | ! | + | ! Name |
! Structure | ! Structure | ||
! width=200px | Notes | ! width=200px | Notes | ||
Revision as of 07:23, 9 March 2010
The nomenclature of steroids is a subset of the nomenclature of natural products.[1]
Definitions
A steroid is a compound (either naturally occurring or artificial) based on the cyclopenta[a]phenanthrene carbon skeleton, partially or completely hydrogenated.[2][3] Steroids usually have methyl groups at C-10 and C-13, and often an alkyl group at C-17 (termed a "side chain"). By extension, one or more bond scissions, ring expansions and/or ring contractions of the skeleton may have occurred.[2][3] Sterols are steroids with a hydroxyl group at C-3.[3]
Fundamental parent structures
| Name | Structure | Notes |
|---|---|---|
| gonane | 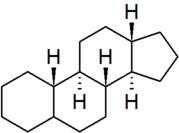
|
|
| estrane | 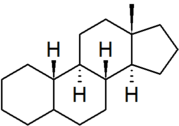
|
"estrane" is the IUPAC preferred spelling; older British texts may spell it as "oestrane" |
| androstane | 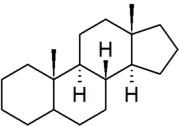
|
|
| pregnane | 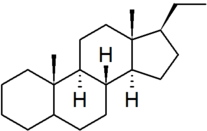
|
|
| cholane | 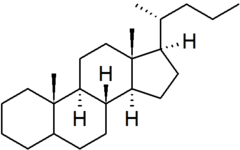
|
|
| cholestane | 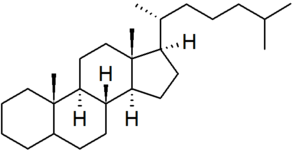
|
|
| ergostane | 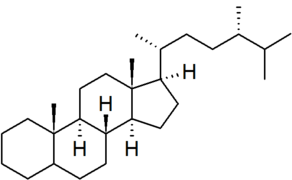
|
|
| campestane | 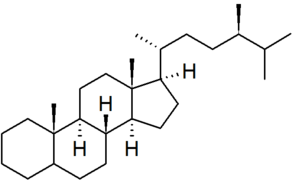
|
|
| poriferastane | 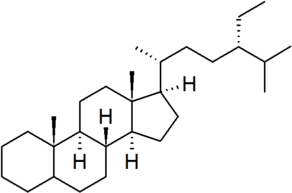
|
|
| stigmastane | 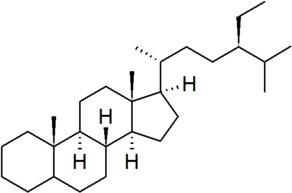
|
|
| gorgostane | 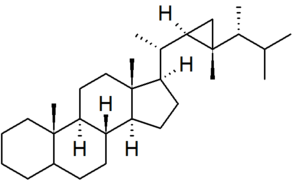
|
numbering of the side chain is non-systematic in CAS nomenclature |
| cardanolide | 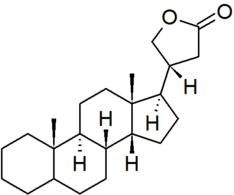
|
|
| bufanolide | 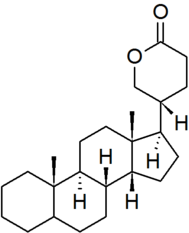
|
|
| furostan | 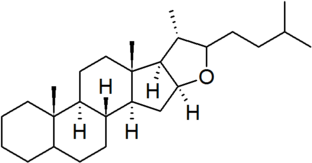
|
configuration at C-22 must be specified for each derivative |
| spirostan | 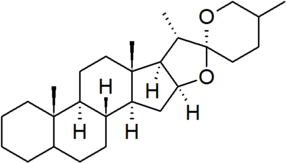
|
configuration at C-25 must be specified for each derivative |
References
- ↑ Revised Section F: Natural Products and Related Compounds (IUPAC Recommendations 1999). Pure Appl. Chem., 71 (4), 587–643. DOI: 10.1351/pac199971040587.
- ↑ 2.0 2.1 steroids, <http://goldbook.iupac.org/S06005.html> (accessed 8 March 2010), Compendium of Chemical Terminology Internet edition; International Union of Pure and Applied Chemistry (IUPAC).
- ↑ 3.0 3.1 3.2 Glossary of class names of organic compounds and reactivity intermediates based on structure (IUPAC Recommendations 1995). Pure Appl. Chem. 1995, 67 (8-9), 1307–75 at 1367. DOI: 10.1351/pac199567081307.
| Error creating thumbnail: Unable to save thumbnail to destination |
This page is currently licensed under the Creative Commons Attribution 3.0 Unported license and any later versions of that license. |