Sulfurous acid
| Sulfurous acid | |
|---|---|
| |

| |
| IUPAC name | Sulfurous acid |
| Identifiers | |
| CAS number | [] |
| Properties | |
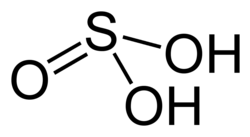
| Chemical formula | H2SO3 |
| Molar mass | 82.07 g/mol |
| Hazards | |
| Material safety data sheet (MSDS) | ICSC 0074 |
| EU index number | 016-011-00-9 |
| EU classification | Corrosive (C) |
| R-phrases | Template:R20, Template:R34 |
| S-phrases | Template:S1/2, Template:S9, Template:S26, Template:S36/37/39, Template:S45 |
| Flash point | Non-flammable |
| Related compounds | |
| Other compounds | Sulfur dioxide Sulfuric acid |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) | |
Sulfurous acid is the chemical compound with the formula H2SO3. There is no evidence that sulfurous acid exists in solution, but the molecule has been detected in the gas phase.[1] The conjugate bases of this elusive acid are, however, common anions, bisulfite (or hydrogensulfite) and sulfite.
Raman spectra of solutions of sulfur dioxide in water show only signals due to the SO2 molecule and the bisulfite ion, HSO3−.[2] The intensities of the signals are consistent with the following equilibrium:
- SO2 + H2O ⇌ HSO3− + H+
- Ka = 1.54 × 10−2; pKa = 1.81.
- SO2 + H2O ⇌ HSO3− + H+
Aqueous solutions of sulfur dioxide, which sometimes are referred to as sulfurous acid are used as reducing agents and as disinfectants, as are solutions of bisulfite and sulfite salts. They are also mild bleaches, and are used for materials which may be damaged by chlorine-containing bleaches.
References
- ↑ D. Sülzle, M. Verhoeven, J. K. Terlouw, H. Schwarz (1988). "Generation and Characterization of Sulfurous Acid (H2SO3) and of Its Radical Cation as Stable Species in the Gas Phase". Angew. Chem. Int. Ed. Engl. 27: 1533–4. doi:10.1002/anie.198815331.
- ↑ Jolly, William L. Modern Inorganic Chemistry, 2nd ed.; McGraw-Hill: New York, 1991. ISBN 0-07-032768-8
See also
- Sulfite
- Sulfuric acid
- Bisulfite
- Carbonic acid, another hypothetical acid used to represent a dissolved gas
| Error creating thumbnail: Unable to save thumbnail to destination | |
This page was originally imported from Wikipedia, specifically this version of the article "Sulfurous acid". Please see the history page on Wikipedia for the original authors. This WikiChem article may have been modified since it was imported. It is licensed under the Creative Commons Attribution–Share Alike 3.0 Unported license. |